Big Idea 6: Any bond or intermolecular attraction that can be formed can be broken. These two processes are in a dynamic competition, sensitive to initial conditions and external perturbations.
Learning Objectives:
Learning objective 6.4: The student can, given a set of initial conditions (concentrations or partial pressures) and the equilibrium constant, K, use the tendency of Q to approach K to predict and justify the prediction as to whether the reaction will proceed toward products or reactants as equilibrium is approached. Learning objective 6.6: The student can, given a set of initial conditions (concentrations or partial pressures) and the equilibrium constant, K, use stoichiometric relationships and the law of mass action (Q equals K at equilibrium) to determine qualitatively and/or quantitatively the conditions at equilibrium for a system involving a single reversible reaction.
Reminders:
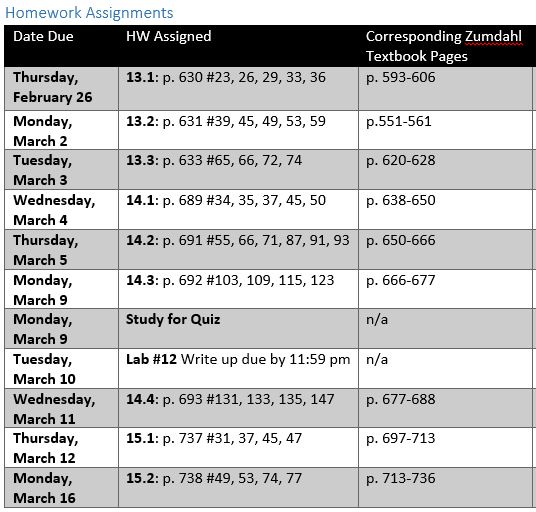
Lab 12 Questions due Wednesday, 3/11 by 11:59 pm
Chapter 13 Quiz Monday, 3/9
Kinetics Exam Tuesday, 2/24
*Quizzes: Any quiz can be retaken once on your time (not in class)
Classwork:
Equilibrium Notes [.PPT]
Club Equilibrium Video
***SMART board slides: Week 23 (2/23-2/27), Week 24 (3/3-3/9)
Exit Ticket: AP Exam Question [.doc]
Equilibrium Worksheet 1 [.doc], 2 [.doc], extra credit [.doc]
LeChatelier's Principle Notes [.PPT]
LeChatelier's Principle Worksheet [.pdf]
Keq and ICE Problems Worksheet [.doc] [.pdf]
Acids Notes [.PPT]
Bases Notes [.PPT]
Salts Notes [.PPT]
Buffers Notes [.PPT]
Acids/Bases in a Nutshell Packet [.pdf], KEY [.pdf]
Exam Study Sheet [.doc]
Learning Objectives:
Learning objective 6.4: The student can, given a set of initial conditions (concentrations or partial pressures) and the equilibrium constant, K, use the tendency of Q to approach K to predict and justify the prediction as to whether the reaction will proceed toward products or reactants as equilibrium is approached. Learning objective 6.6: The student can, given a set of initial conditions (concentrations or partial pressures) and the equilibrium constant, K, use stoichiometric relationships and the law of mass action (Q equals K at equilibrium) to determine qualitatively and/or quantitatively the conditions at equilibrium for a system involving a single reversible reaction.
Reminders:
Lab 12 Questions due Wednesday, 3/11 by 11:59 pm
Chapter 13 Quiz Monday, 3/9
Kinetics Exam Tuesday, 2/24
*Quizzes: Any quiz can be retaken once on your time (not in class)
Classwork:
Equilibrium Notes [.PPT]
Club Equilibrium Video
***SMART board slides: Week 23 (2/23-2/27), Week 24 (3/3-3/9)
Exit Ticket: AP Exam Question [.doc]
Equilibrium Worksheet 1 [.doc], 2 [.doc], extra credit [.doc]
LeChatelier's Principle Notes [.PPT]
LeChatelier's Principle Worksheet [.pdf]
Keq and ICE Problems Worksheet [.doc] [.pdf]
Acids Notes [.PPT]
Bases Notes [.PPT]
Salts Notes [.PPT]
Buffers Notes [.PPT]
Acids/Bases in a Nutshell Packet [.pdf], KEY [.pdf]
Exam Study Sheet [.doc]
 RSS Feed
RSS Feed
