Big Idea: The laws of thermodynamics describe the essential role of energy and explain and predict the direction of changes in matter.
Learning Objectives:
Learning objective 5.4 The student is able to use conservation of energy to relate the magnitudes of the energy changes occurring in two or more interacting systems, including identification of the systems, the type (heat versus work), or the direction of energy flow.
Learning objective 5.5 The student is able to use conservation of energy to relate the magnitudes of the energy changes when two nonreacting substances are mixed or brought into contact with one another.
Learning objective 5.6 The student is able to use calculations or estimations to relate energy changes associated with heating/cooling a substance to the heat capacity, relate energy changes associated with a phase transition to the enthalpy of fusion/vaporization, relate energy changes associated with a chemical reaction to the enthalpy of the reaction, and relate energy changes to PΔV work.
Learning objective 5.7 The student is able to design and/or interpret the results of an experiment in which calorimetry is used to determine the change in enthalpy of a chemical process (heating/cooling, phase transition, or chemical reaction) at constant pressure.
Reminders:
Late policy: 2nd offense is a 20 minute lunch detention
Assignment Tardiness: All assignments for a unit are ultimately due by the unit test before points are taken off for tardiness
Quizzes: Any quiz can be retaken once on your time (not in class)
Classwork:
Mon 1/5 (A)
1 – Review Gases HW from break
2 – Grade and Review Gases Take Home Quiz from break
HW - n/a
Tues 1/6 (B)
Intro to Thermochemistry & Solids/Liquids Unit
1 - Stations: Internal Energy, Heat, Work [.PPT], Capture Sheet [.doc]
HW: Textbook HW
Wed 1/7 (C)
1 - Calorimetry Notes [.PPT]
1/2 - Calorimetry Activity Packet
HW: Calorimetry Pre-Lab (Questions and Data Tables)
Thurs 1/8 (D)
1 – LAB #10: Calorimetry
HW: Textbook HW
Fri 1/9 (A)
1– LAB #10 continued
HW: Textbook HW, Formal Lab Write Up due next Friday (not Thurs)
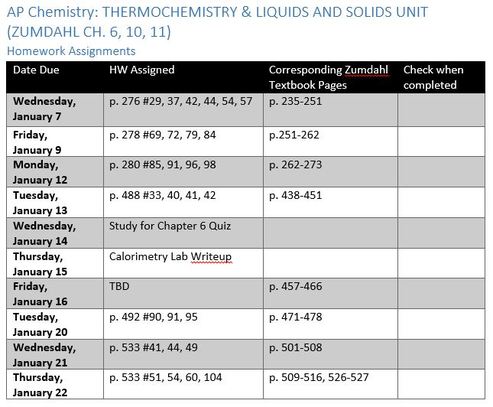
Homework (assignments for rest of marking period): [.doc]
Learning Objectives:
Learning objective 5.4 The student is able to use conservation of energy to relate the magnitudes of the energy changes occurring in two or more interacting systems, including identification of the systems, the type (heat versus work), or the direction of energy flow.
Learning objective 5.5 The student is able to use conservation of energy to relate the magnitudes of the energy changes when two nonreacting substances are mixed or brought into contact with one another.
Learning objective 5.6 The student is able to use calculations or estimations to relate energy changes associated with heating/cooling a substance to the heat capacity, relate energy changes associated with a phase transition to the enthalpy of fusion/vaporization, relate energy changes associated with a chemical reaction to the enthalpy of the reaction, and relate energy changes to PΔV work.
Learning objective 5.7 The student is able to design and/or interpret the results of an experiment in which calorimetry is used to determine the change in enthalpy of a chemical process (heating/cooling, phase transition, or chemical reaction) at constant pressure.
Reminders:
Late policy: 2nd offense is a 20 minute lunch detention
Assignment Tardiness: All assignments for a unit are ultimately due by the unit test before points are taken off for tardiness
Quizzes: Any quiz can be retaken once on your time (not in class)
Classwork:
Mon 1/5 (A)
1 – Review Gases HW from break
2 – Grade and Review Gases Take Home Quiz from break
HW - n/a
Tues 1/6 (B)
Intro to Thermochemistry & Solids/Liquids Unit
1 - Stations: Internal Energy, Heat, Work [.PPT], Capture Sheet [.doc]
HW: Textbook HW
Wed 1/7 (C)
1 - Calorimetry Notes [.PPT]
1/2 - Calorimetry Activity Packet
HW: Calorimetry Pre-Lab (Questions and Data Tables)
Thurs 1/8 (D)
1 – LAB #10: Calorimetry
HW: Textbook HW
Fri 1/9 (A)
1– LAB #10 continued
HW: Textbook HW, Formal Lab Write Up due next Friday (not Thurs)
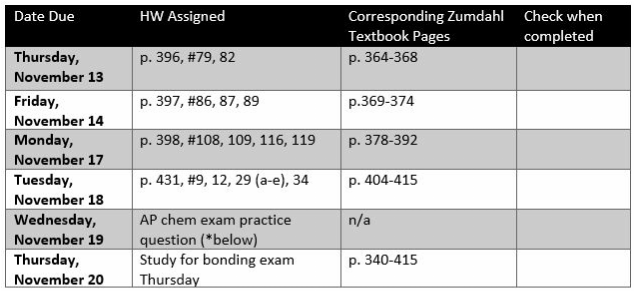
Homework (assignments for rest of marking period): [.doc]

 RSS Feed
RSS Feed
